|
Read More: What foods can cause hallucinations? Is trehalose a non reducing sugar? … It is the opposite in an l-configuration furanose. A furanose ring structure consists of four carbon and one oxygen atom with the anomeric carbon to the right of the oxygen. The furanose ring is a cyclic hemiacetal of an aldopentose or a cyclic hemiketal of a ketohexose. It is the dominant structural isoform in crystalline fructose. What is the functional group of alpha D Fructofuranose?ĬHEBI:37720 – -D-fructofuranose -D-fructofuranose is a Functional Parent ofĪlpha-D-fructofuranose 1,6-bisphosphate Mass : 340.11568 Formula : C6H14O12P2Īlpha-D-fructuronic acid Mass : 194.13940 Formula : C6H10O7īeta-D-fructopyranose is the six-member ring form of fructose: a simple ketonic monosaccharide found in many plants and often bonded to glucose to form the disaccharide sucrose. Therefore, some health professionals recommend fructose as a safe sweetener for people with type 2 diabetes ( 3 ). Unlike glucose, fructose causes a low rise in blood sugar levels. Why is carbohydrate called carbohydrate?Įtymology: Carbohydrates are called carbohydrates because the carbon, hydrogen and oxygen they contain are usually in the proportion to form water with the general formula Cn(H2O)n. … Thus, although the ketose fructose is not strictly a reducing sugar, it is an alpha-hydroxy-ketone and gives a positive test because the base in the reagent converts it into the aldoses glucose and mannose. Such tests that use this reagent are called the Benedict’s tests. Why does fructose give a positive Benedict test? The sugar is now in a form that can follow the normal glycolytic pathway. The newly formed mannose-6-phosphate is then isomerized into fructose-6-phosphate by the enzyme phosphomannose isomerase. How does mannose enter glycolysis?Įxplanation: Mannose enters glycolysis by first being phosphorylated by hexokinase. Naturally occurring fructose is (-) isomer with D-configuration. its molecule possesses one aldehyde group and five hydroxyl groups is primary and rest four are secondary in nature. Read More: What is Gan Cao used for? How many alcohol groups are in a Fructofuranose?Īnswer: It is bifunctional compound i.e. Its systematic name is -D-galactopyranosyl-(14)-D-glucose. Lactose is a disaccharide derived from the condensation of galactose and glucose, which form a -14 glycosidic linkage. … D-Glucopyranose having alpha-configuration at the anomeric centre. These are monosaccharides in which the sugar unit is a is a six-carbon containing moeity. What is Alpha D-Glucopyranose?Īlpha-D-Glucose, also known as alpha-dextrose or alpha-D-GLC, belongs to the class of organic compounds known as hexoses. It is an enantiomer of a beta-L-galactose. It has a role as an epitope and a mouse metabolite. What is beta D galactose?īeta-D-galactose is a D-galactopyranose having beta-configuration at the anomeric centre. The product is called fructofuranose in analogy with the five-membered ring of furan.  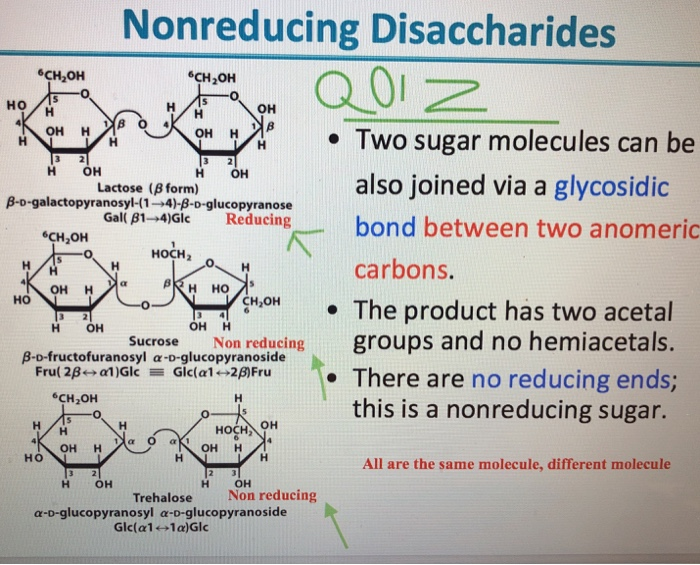 
In fructose, the OH group at C5 can attack the C=O. It is formed by the reaction of acetone with an alcohol. Example 2: -D-Fructofuranose and -D-fructofuranose are anomers. Example 1: -D-Glucopyranose and -D-glucopyranose are anomers. The epimeric carbon in anomers are known as anomeric carbon or anomeric center. Which carbon is anomeric in Fructofuranose? is that fructose is (carbohydrate) a monosaccharide ketose sugar, formula c 6h 12o 6 while fructofuranose is (carbohydrate) the furanose form of fructose. In contextcarbohydratelang=en terms the difference between fructose and fructofuranose. What is the difference between fructose and Fructofuranose? … alpha-D-fructofuranose is a reducing sugar. This means that the OH group from the aldehyde of glucose is not free–not a reducing sugar. The methyl glycoside is the ether bond between the two glucose residues of cellobiose. Since alpha-D-fructofuranose is a monosaccharide, it is a reducing agent. What is the structure of Fructofuranose?ĭ-fructofuranose D-Arabino-hexulose D-fructofuranose (closed ring structure) 1 beta-D-Fructose More… It is an enantiomer of a beta-L-fructofuranose. Beta-D-fructofuranose is a D-fructofuranose. What is a beta D-fructofuranose?ĭescription. As is evident from its structure (its anomeric carbon atoms are involved in glycosidic bonds), it is a non-reducing sugar.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
